Nanoplastics smaller than 400 nanometers can penetrate biological barriers and accumulate in tissues, a phenomenon sparking widespread concern in toxicology and public health. Studies show these ultra-small particles may induce multi-systemic toxicity, including brain and nervous system damage, cardiovascular dysfunction, reproductive risks, as well as cellular and gastrointestinal toxicity. Finding trace amounts of nanoplastics in complex, high-viscosity matrices like blood or serum—which are packed with proteins and inorganic salts—is akin to finding a needle in a haystack, and traditional analytical tools often fall short. The excessively long sample preparation times and a lack of detection sensitivity for ultra-micro particles represent massive technical roadblocks for current environmental toxicology and clinical assessments (as shown in Figure 1). 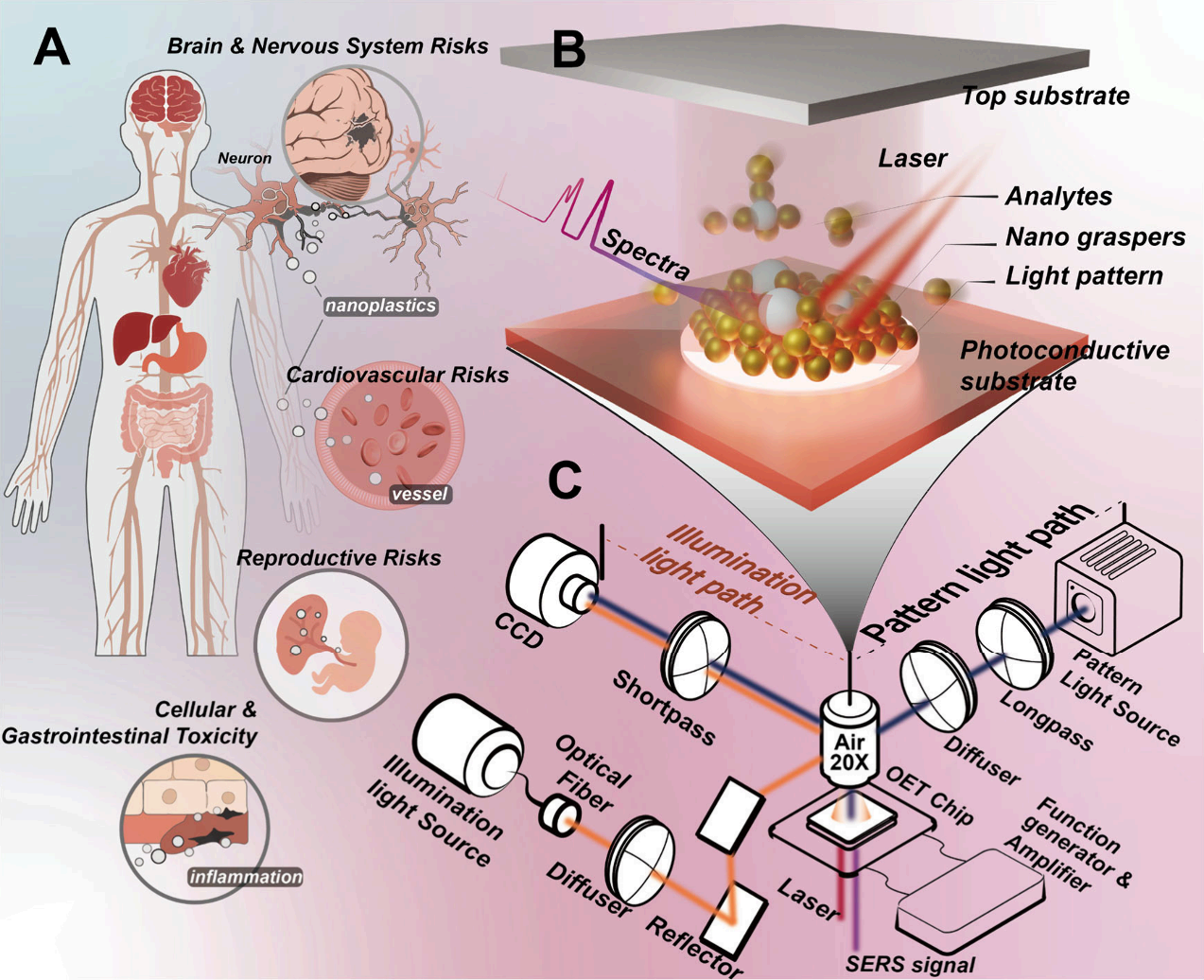
Figure 1. (A) Schematic illustration of the impacts of nanoplastics on health. (B) Schematic illustration of the OET chip, where light patterns guide the manipulation of nanograspers to capture and enrich target analytes. (C) Optical setup and system conffguration of the OET-NGP platform for nanoparticle manipulation and SERS detection.
Recently, a team led by Professor Shuailong Zhang and Associate Researcher Rongxin Fu from the Beijing Institute of Technology, alongside various scholars, published highly inspiring work in ACS Nano. They designed an Optoelectronic Tweezer (OET) assisted "nanograsper" system. This system leverages structured light to guide the synergistic effects of light-induced AC electroosmosis (LACE) and dielectrophoresis (DEP) to dynamically assemble gold nanoparticles into "nanograspers" in the liquid phase. It can actively grasp and encapsulate target nanoplastics in approximately 10 seconds, rapidly enriching analytes while forming highly uniform hotspots to generate intense Surface-Enhanced Raman Scattering (SERS) signals. This work brilliantly resolves the challenge of detecting trace macromolecules and particulate matter in complex biological fluids.
The Technical Trade-off in Finding the Perfect SERS Substrate
In nanoscale analyte detection, SERS technology is widely favored for its molecular-level fingerprint recognition capabilities. However, a core dilemma persists in this field: how to obtain SERS hotspots that are simultaneously highly uniform and possess immense signal amplification power.
Traditional SERS platforms are generally split into two camps. The first is colloidal systems, where metal nanoparticles are suspended in solution; this is cost-effective and easy for high-throughput operations, but the random dispersion often leads to highly unstable signals, uncontrollable aggregation at low concentrations, or unwanted surface adsorption. The second type includes solid structure substrates based on micro/nanofabrication, which offer clearly defined hotspots and excellent signal reproducibility. However, solid substrates are complex and time-consuming to manufacture, and their static geometric layouts lack the flexibility needed to track moving analytes or dynamically adjust to changing sample conditions.
Researchers urgently needed a next-generation detection platform that fuses the flexibility of liquid systems with the stability of solid state systems.
Decoding the Mechanism: The Perfect Synergy of Invisible Robotic Arms and Programmable Dynamic Matrices
To overcome these limitations, the research team abandoned traditional static substrate preparation and developed a highly reconfigurable dynamic capture system. They introduced Optoelectronic Tweezer (OET) technology into their experiments, brilliantly utilizing the synergy of LACE and DEP forces to achieve precise manipulation of metal nanoparticles in a liquid phase.
The core hardware for the experiment relied on Optoseeker Biotech's OptoBot® 500 Optoelectronic Tweezer System. This platform integrates a high-precision, DMD-based structured light projection module with a flexible electric field control unit. Researchers uniformly dispersed free 60 nm gold nanoparticles (AuNPs) in the liquid layer of the OET chip. When the system applied a 15 kHz AC bias voltage and projected specific light patterns downwards onto the photoconductive film (a-Si:H) via the OptoBot® 500, the conductivity of the illuminated area rapidly changed, causing local electric field redistribution (as shown in Figure 2).
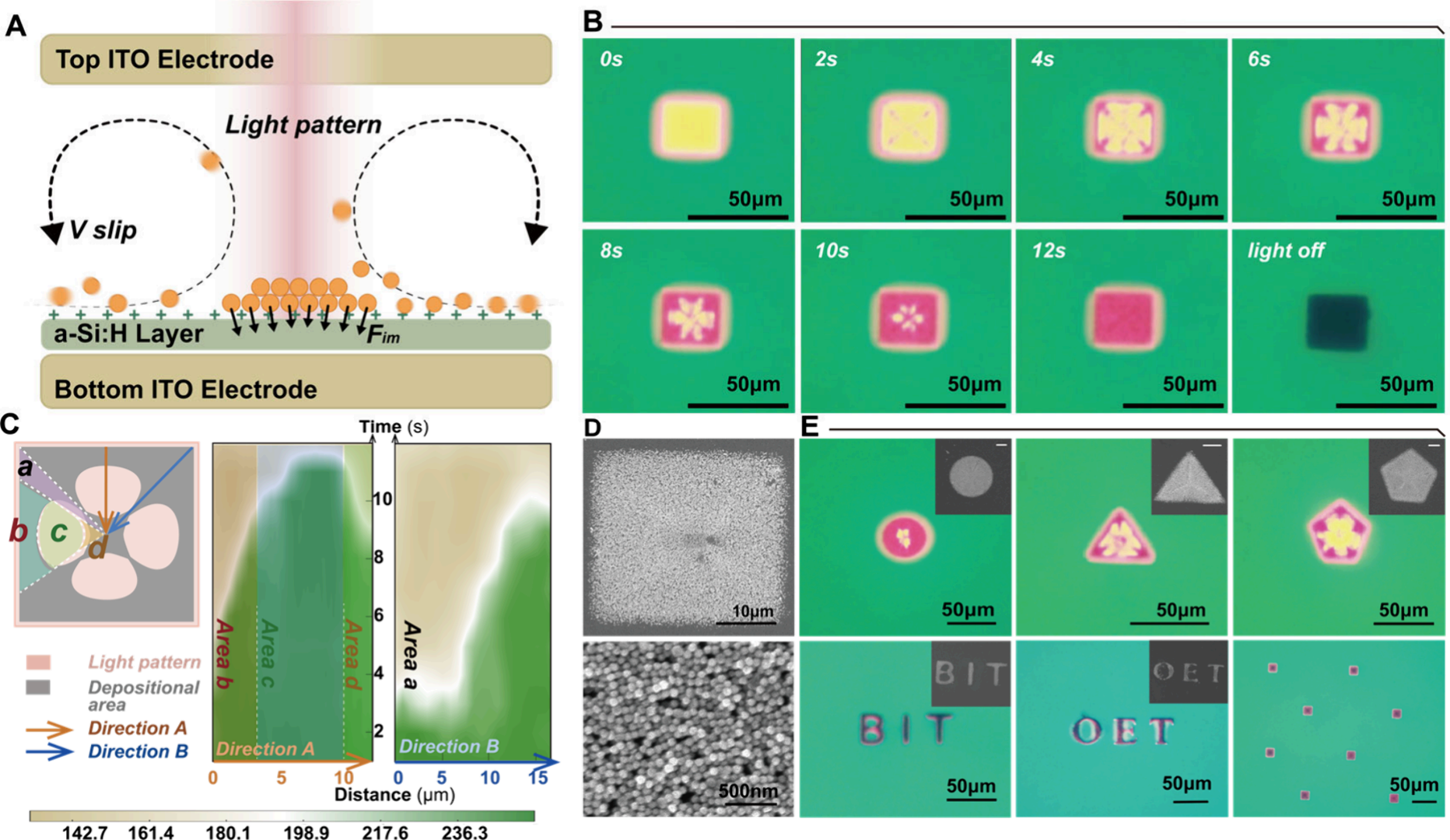
Figure 2. (A) Cross-sectional schematic of the OET chip structure and its operating principle, illustrating the manipulation of nanograspers via lightinduced forces. (B) Time-sequence images illustrating the aggregation of AuNPs under a rectangular light pattern, followed by their immobilization on the substrate. (C) Time-dependent grayscale heatmap generated based on pixel intensity analysis, illustrating the evolution of nanoparticle accumulation along two directions within the illuminated region: the perpendicular direction along the edge (Direction A) and the diagonal direction (Direction B). A schematic inset divides the deposition process into ffve regions (Area A to D), indicating the temporal sequence of nanoparticle accumulation. (D) Corresponding scanning electron microscopy (SEM) images of the immobilized AuNP structures, conffrming the high-ffdelity pattern formation. (E) Demonstration of high-precision, ffexible assembly into arbitrary shapes via light-controlled manipulation. The SEM image illustrates the assembled structure (scale bar: 10 μm)
This triggers a powerful physical effect: light-induced electroosmotic flow creates fluid convection on the periphery of the illuminated area, continuously transporting distant free gold nanoparticles and target analytes to the boundary; a subsequently powerful dielectrophoretic force acts like a nail, firmly fixing these particles to the underlying substrate. Under a microscope, free nanoparticles can be clearly seen rushing toward the geometric center and vertices under the guidance of the light pattern, ultimately arranging into densely packed, designated shapes. This assembly process, spreading from the edges to the center, resembles a clenching fist that tightly envelops the surrounding nanoplastics, instantaneously constructing an abundance of SERS hotspots with intense inter-particle coupling effects.
Breakthrough Results: Leaping from Pure Water Media to Real Serum
This dynamic assembly strategy demonstrated outstanding data performance in both detection speed and sensitivity. Experimental records show that it takes the system only about 12 seconds to complete the assembly of a dense, 30-square-micrometer nanoparticle array on the chip.
Using Rhodamine B to verify ultimate performance, the calibration curve revealed a Limit of Detection (LOD) as low as 4.43 × 10-8 M. More crucially, the signal consistency was remarkably high: within a single "nanograsper" structure, the relative standard deviation (RSD) of the characteristic peak intensity at 1505 cm-1 was only 8.11%; across five independently generated structures, the batch-to-batch RSD was as low as 6.89%. This data completely meets the stringent signal fluctuation requirements of high-quality commercial SERS substrates (as shown in Figure 3).
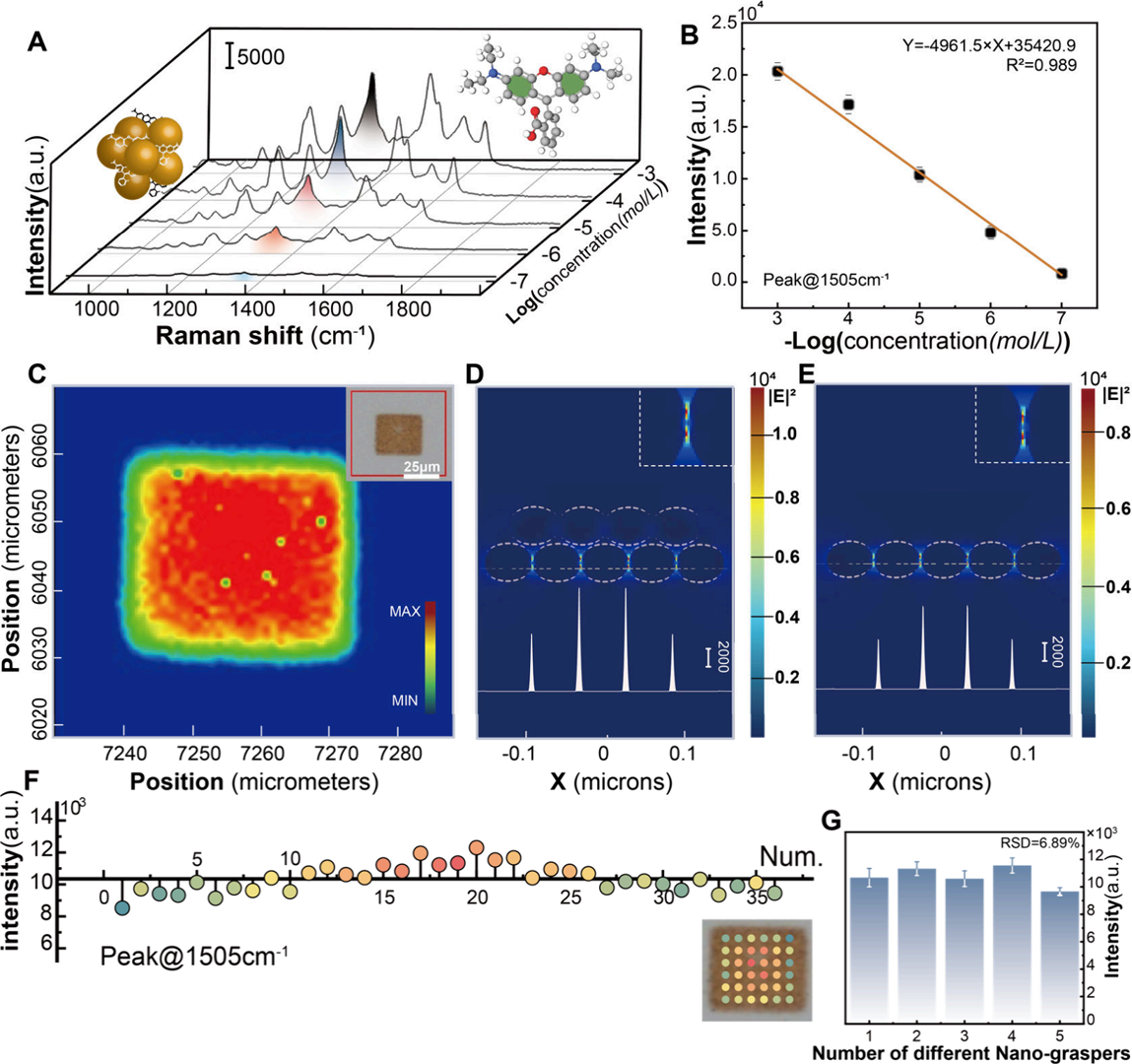
Figure 3. (A) Raman spectra of nanograspers capturing Rhodamine B from solution, with concentrations ranging from 10−3 M to 10−7 M. (B) Linear fft of the logarithmic concentration of Rhodamine B versus peak intensity at 1505 cm−1 . (C) Raman mapping of nanograsper deposition on the substrate, demonstrating uniformity. (D,E) Comparison of FDTD simulations of electric ffeld intensity between deposited multilayer nanograsper structure (D) and single-layer assembled structure (E), both showing electric ffeld distribution with a 30 nm height cross-section. A 21% intensity increase is observed in (D). (F) Raman peak intensity at 1505 cm−1 measured at different locations within a single nanograsper pattern, showing an RSD of 8.11%. (G) SERS peak intensity comparison from ffve different nanograspers, yielding an RSD of 6.89%
For representative nanoplastics like polystyrene (PS) and polyethylene terephthalate (PET), the platform reached LODs of 5.6 μg/mL and 8.9 μg/mL respectively in pure water environments. The most compelling breakthrough of this work, however, is its direct detection capability in real human serum samples (as shown in Figure 4).
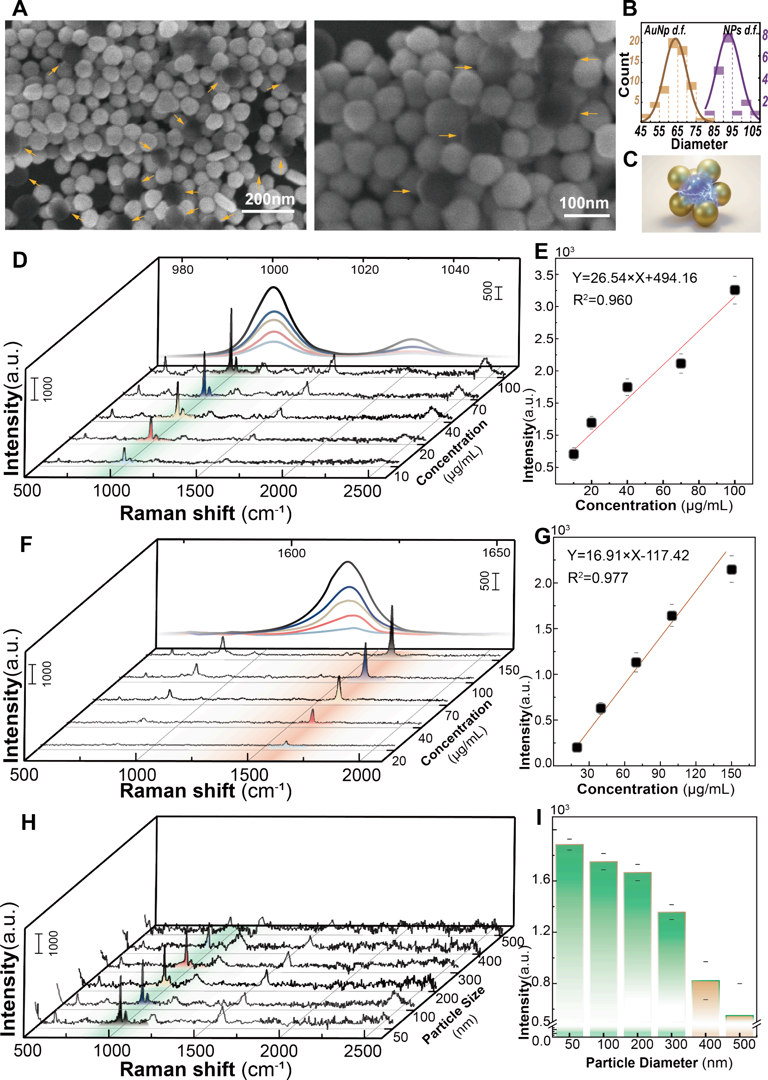
Figure 4. SERS-based detection of nanoplastics using the OET-NGP. (A) SEM images of gold nanoparticle-assembled nanograspers encapsulating nanoplastic beads. (B) Size distribution of gold nanoparticles and nanoplastic beads. (C) Schematic illustration of the SERS signal generation process by a nanograsper encapsulating a nanoplastic particle. (D) SERS spectra of PS nanoplastic beads at varying concentrations (10, 20, 40, 70, and 100 μg/ mL). (E) Calibration curve of the peak intensity at 1003 cm−1 as a function of PS bead concentration. (F) SERS spectra of PET nanoplastic beads at different concentrations (20, 40, 70, 100, and 150 μg/mL). (G) Calibration curve of the peak intensity at 1614 cm−1 as a function of PET bead concentration, indicating the detection sensitivity of the platform. (H) SERS spectra of PS nanoplastics with different particle sizes. (I) Comparison of the peak intensity at 1003 cm−1 for PS nanoplastics of different sizes.
Serum features extremely high viscosity, complex ionic environments, and massive background biomacromolecules—factors that easily disrupt nanoparticle electrokinetic behavior and cause non-specific precipitation. The research team developed a two-step voltage enrichment strategy on the OptoBot® 500 platform. The first phase applies a lower voltage of 15 V, utilizing the size selectivity of low-Reynolds-number hydrodynamics. Relying mainly on AC electroosmotic flow, it gently gathers the nanograspers and nanoplastics into the illuminated area, avoiding premature deposition of macromolecular impurities. Once a highly concentrated local enrichment zone is formed, the voltage rapidly switches to 25 V, utilizing intensified dielectrophoretic force to firmly immobilize the enriched complex.
Experimental validation showed that background signals from biomolecules like uric acid remained at a highly stable baseline, and interferents like Bovine Serum Albumin (BSA) and DNA had virtually no negative impact. Direct comparison against the industry-standard pyrolysis-gas chromatography-mass spectrometry (pyGC-MS) clearly proved the method's quantitative accuracy: in the low concentration range of 10 to 20 μg/mL, pyGC-MS exhibited significant quantitative deviation due to severe serum matrix interference, whereas the OET-based "nanograsper" SERS method maintained excellent linear response and signal consistency (as shown in Figure 5).
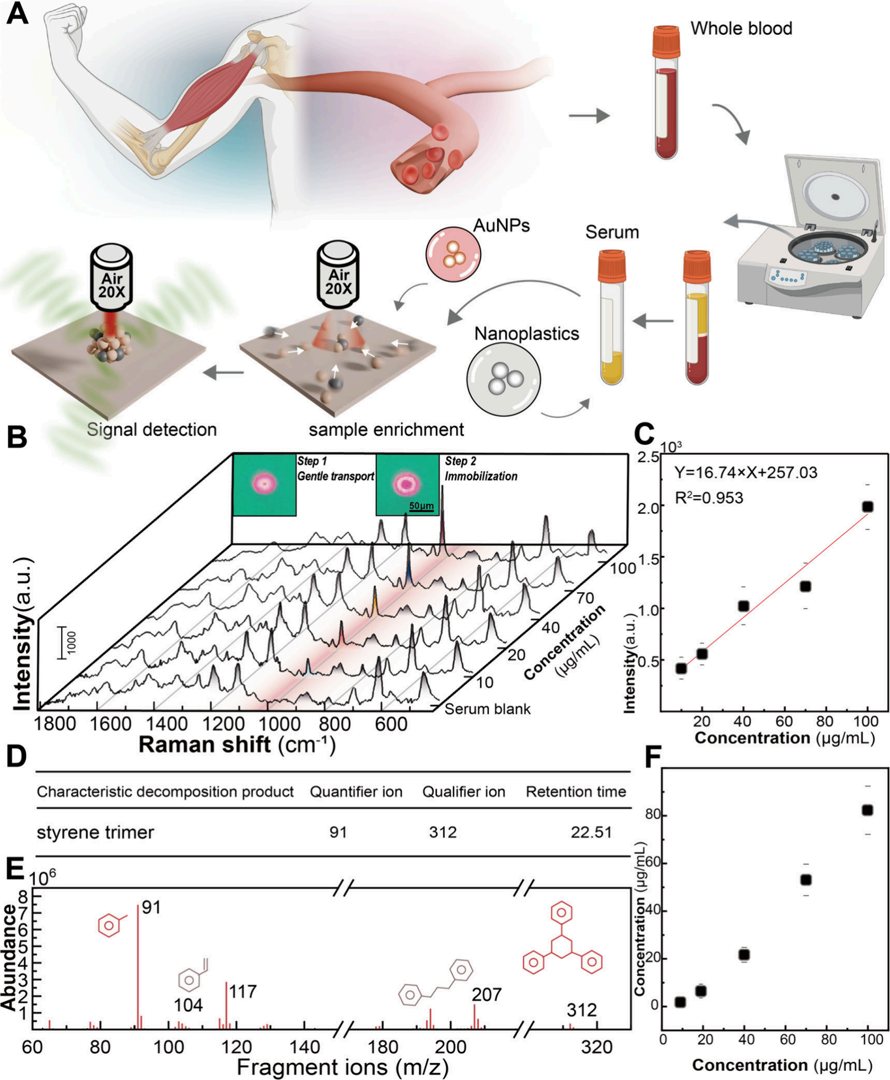
Figure 5. SERS-based detection of nanoplastics in serum using the OET-NGP platform. (A) Schematic of the sample preparation and detection workffow. (B) Illustration of the enrichment process of nanograsper−nanoplastic complexes in serum under a two-step voltage, along with corresponding SERS spectra at varying nanoplastic concentrations. (C) Calibration curve of peak intensity versus nanoplastic concentration, showing good linearity. (D) Secondary mass spectrum of the selected signature ion (SSS), conffrming the molecular structure of the nanoplastic component. (E) PyGC−MS chromatogram of the serum sample spiked with nanoplastics, showing the characteristic ions and their corresponding retention times for qualitative identiffcation. (F) Quantitative calibration curve of the selected ion signal intensity versus nanoplastic concentration, supporting the SERS quantiffcation results.
Methodological Implications: From Passive Waiting to Active Enrichment
The technical route demonstrated here provides a brand-new optimization perspective for the biochemical sensing field.
The vast majority of highly sensitive optical detection methods passively wait for target molecules to diffuse to the sensor surface. At extremely low concentrations, the collision probability driven by Brownian motion drops exponentially, severely prolonging detection cycles. This research achieves true "active enrichment" using the optoelectronic microfluidic platform. Powerful local fluid vortices forcefully pull distant targets into the detection zone, completely shattering the physical bottlenecks of diffusion limits.
Because light patterns can be perfectly defined in real-time via software, researchers easily projected triangle, hexagon, and even "BIT" letter-shaped SERS arrays during the experiment. This high degree of spatial programmability means sensors can be "printed on demand" anywhere on the microfluidic chip, tailored for specific microenvironments of different regions, to complete multi-site parallel measurements.
Naturally, this technical solution has specific applicability boundaries. Research shows that when target plastic particles exceed 400 nm in diameter, the Raman signal intensity drops significantly alongside higher fluctuation. This is primarily due to a size mismatch between micron-scale large particles and 60 nm gold particles; large particles struggle to be effectively encapsulated into the tight gaps constructed by smaller particles, lowering hotspot density. This physical limitation (size matching effect) clarifies the technology's optimal application scenario: targeting the detection of sub-400 nm microparticles, which hold higher penetration capability and greater toxicological significance.
A New Window for Clinical Diagnostics and Environmental Toxicology
Whether it's tracking the metabolism of microplastics in the human body, or rapidly analyzing trace exosomes and cell-free DNA in liquid biopsies, direct detection in complex biological fluids without pre-treatment remains the pinnacle of life science instrumentation development.
By combining high-precision light-control equipment with nano-assembly technology, the research team successfully compressed SERS substrate preparation, target enrichment, and signal readout into an astonishing ten-plus seconds. This plug-and-play dynamic sensing philosophy drastically simplifies experimental workflows and reduces consumable costs. With further refinement of targeted separation strategies, this "nanograsper" platform, powered by Optoelectronic Microfluidics, is poised to become a standard experimental tool for clinical blood screening and environmental pollutant toxicity assessment, offering a highly commercially viable shortcut for next-generation Point-of-Care Testing (POCT) and high-throughput bioanalysis.
Featured Product Integration:
Optoseeker Biotech's OptoBot® 500 Optoelectronic Tweezer System utilizes Digital Micromirror Device (DMD) projection technology and integrates waveform generation capabilities. It can complete the manipulation of micro/nano targets (metal particles, dielectric particles, nanomaterials, cells, etc.) without the need for additional components. Integrated with the OptoMind® 500 operating software, it enables graphical editing and motion control of micro/nano targets through intuitive software commands.
Click to learn more about the product
Original Publication:
Light-Programmable Nanograspers for Rapid Nanoplastics Detection in Biological Fluids
DOI.org/10.1021/acsnano.5c20989







